Clinical trial of ChitoCare® medical Wound Healing Gel published in the British Medical Journal
New clinical study shows ChitoCare® medical Wound Healing Gel increases the healing rate of chronic diabetic foot ulcers.
Siglufjörður, Iceland - [4 July 2024] – Primex Iceland, a leading marine biotechnology company focused on innovative wound healing solutions, announces the publication of full results from a clinical trial of its proprietary ChitoCare® medical Wound Healing Gel in the British Medical Journal (BMJ) Open Diabetes Research & Care.
The trial investigated the treatment of diabetic foot ulcers (DFUs), which are one of the leading causes of lower-limb amputations worldwide. Current standard-of-care (SOC) may not properly address the adequate healing and recurrence of DFUs.
The study reported that at the end of a 10-week treatment period, more patients had fully healed, and 50% and 75% wound closure was significantly higher, using the chitosan-based Wound Healing Gel in addition to SOC compared to SOC plus a placebo gel. Patients treated with the Wound Healing Gel experienced a significant reduction in wound surface area compared to placebo at the end of the study. In addition, wound state was significantly improved in patients using the Wound Healing Gel vs placebo.
The Wound Healing Gel proved to be a safe and effective adjuvant to standard-of-care therapy for the treatment of DFUs. One of the advantages of the Wound Healing Gel is its ease of application, which enables home use, and thus eliminates the need for frequent clinic visits. This could considerably reduce DFU treatment costs while also improving patient outcomes.
“We are thrilled with the positive results of this clinical trial,” said Sigríður Vigfúsdóttir, CEO of Primex Iceland. “These findings provide strong evidence that our Wound Healing Gel has the potential to significantly improve the lives of patients suffering from chronic wounds such as diabetic foot ulcers.”
Primex thanks Principal Investigator Dr Maja Navodnik Preložnik for her conduct of the study, collaborators Vizera d.o.o and the University of Ljubljana for their involvement, and all patients for their participation in the trial. Special thanks also go to Vilma Urbančič Rovan for her support and to all authors of the publication. The study was supported by two grants received by Primex Iceland from the AVS Research Fund of The Ministry of Fisheries in Iceland.
Additional studies are ongoing and planned to supplement the results of this trial.
CHITOWOUND study details
The study was a prospective, randomized, multicentre, placebo-controlled trial which compared the safety and efficacy of ChitoCare® medical Wound Healing Gel in addition to SOC vs placebo gel plus SOC in the treatment of chronic, hard-to-heal, diabetic foot ulcers across multiple sites in Slovenia and Croatia (CHITOWOUND study, NCT04178525).
Patients with diabetes and a non-healing, uninfected DFU, or a post-amputation wound that was present for at least 4 weeks, were invited to participate. All patients must have had at least 4-weeks of SOC treatment consisting of off-loading therapy. Participants who did not have this SOC treatment underwent a 4-week run-in period. The study included a 10-week treatment period and 4-week follow-up.
Detailed study results
42 patients were included in the intention-to-treat (ITT) population, with 18 patients randomised in the ChitoCare arm and 24 in the placebo arm.
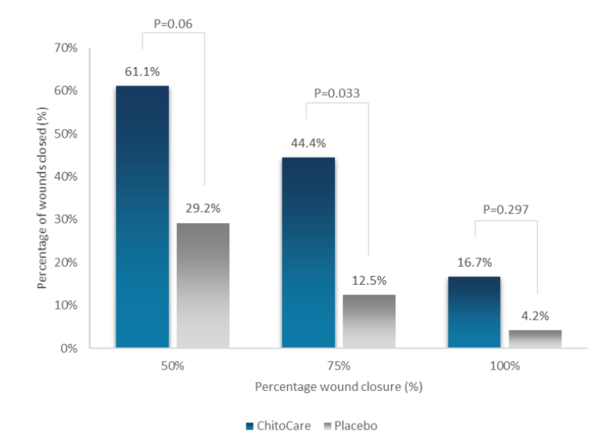
More patients (16.7%) achieved complete wound closure in the ChitoCare arm compared to the placebo arm (4.2%) at week 10 (p=0.297). Additionally, the ChitoCare arm showed a 4.44-fold higher likelihood to completely heal over 10 weeks compared with the placebo arm (p=0.158).
At week 10, 75% wound closure was higher in the ChitoCare arm (44.4%) compared to placebo (12.5%) (p=0.033). A similar trend was observed for 50% wound closure (61.1% vs 29.2%, p=0.060).
The ChitoCare arm showed a statistically significant 4.62-fold higher likelihood to achieve 75% wound closure at 10 weeks compared with the placebo arm (p=0.012).
Additionally, the ChitoCare arm experienced a statistically significant reduction in wound surface area (92.0% vs 37.0%, p=0.008) and improvement in wound state as measured by the Bates-Jensen Wound Assessment Tool (BWAT) (24 vs 20 points, p=0.018). The relative improvement from the baseline wound state was also significantly higher in the ChitoCare arm (median 29.8%) compared with the placebo arm (median 3.6%, p=0.010).
About Primex
Primex is an Icelandic marine biotechnology company specialising in the sustainable production of high-quality chitosan. Primex operates out of Siglufjörður in the North of Iceland and leverages the clean waters of the North Atlantic for its raw material. Primex's state-of-the-art facility produces chitosan and related products under the brand names of ChitoCare®, ChitoClear®, LipoSan Ultra®, and SeaKlear®.
Primex's chitosan is sourced from the discarded shells of the Pandalus borealis shrimp, which is usually considered a waste product from the local fishing industry. The collection and manufacturing of Primex's products uses processes that contribute to a more circular and sustainable economy in the Arctic.
Primex is the producer of three medical devices, ChitoCare® medical Wound Healing Gel, ChitoCare® medical Healing Spray and ChitoCare® medical Scar Healing Gel. These devices are designed to assist wound and/or scar healing and protect the skin. ChitoCare® medical are CE marked Class III medical devices in accordance with the European Directive 93/42/EEC on Medical Devices.
About ChitoCare® medical Wound Healing Gel

ChitoCare® medical Wound Healing Gel is a topical gel for acute and chronic wounds and other skin conditions. It protects the skin and wounds against infections, helps reduce bleeding, scarring, and pain due to its cooling effect. It soothes the symptoms associated with various skin conditions, such as itching and redness, promoting the natural skin repair process. It can also effectively reduce acute and chronic wounds. It can be applied to any skin area or wound, forming a thin, transparent, and breathable film.

